-
PDF
- Split View
-
Views
-
Cite
Cite
R. K. Gherardi, M. Coquet, P. Cherin, L. Belec, P. Moretto, P. A. Dreyfus, J.-F. Pellissier, P. Chariot, F.-J. Authier, Macrophagic myofasciitis lesions assess long-term persistence of vaccine-derived aluminium hydroxide in muscle, Brain, Volume 124, Issue 9, September 2001, Pages 1821–1831, https://doi.org/10.1093/brain/124.9.1821
Close - Share Icon Share
Abstract
Macrophagic myofasciitis (MMF) is an emerging condition of unknown cause, detected in patients with diffuse arthromyalgias and fatigue, and characterized by muscle infiltration by granular periodic acid–Schiff's reagent-positive macrophages and lymphocytes. Intracytoplasmic inclusions have been observed in macrophages of some patients. To assess their significance, electron microscopy was performed in 40 consecutive cases and chemical analysis was done by microanalysis and atomic absorption spectrometry. Inclusions were constantly detected and corresponded to aluminium hydroxide, an immunostimulatory compound frequently used as a vaccine adjuvant. A lymphocytic component was constantly observed in MMF lesions. Serological tests were compatible with exposure to aluminium hydroxide-containing vaccines. History analysis revealed that 50 out of 50 patients had received vaccines against hepatitis B virus (86%), hepatitis A virus (19%) or tetanus toxoid (58%), 3–96 months (median 36 months) before biopsy. Diffuse myalgias were more frequent in patients with than without an MMF lesion at deltoid muscle biopsy (P < 0.0001). Myalgia onset was subsequent to the vaccination (median 11 months) in 94% of patients. MMF lesion was experimentally reproduced in rats. We conclude that the MMF lesion is secondary to intramuscular injection of aluminium hydroxide-containing vaccines, shows both long-term persistence of aluminium hydroxide and an ongoing local immune reaction, and is detected in patients with systemic symptoms which appeared subsequently to vaccination.
Introduction
Macrophagic myofasciitis (MMF) is a recently recognized entity, emerging in 1993 from France (Gherardi et al., 1998). Affected patients have diffuse steroid-responsive arthromyalgias and marked fatigue as their main clinical symptoms. Deltoid muscle biopsy shows stereotypical perimuscular infiltration by large macrophages with a finely granular periodic acid–Schiff's reagent (PAS)-positive content intermingled with lymphocytic infiltrates, and inconspicuous muscle fibre damage. MMF is distinct from idiopathic and sarcoid-like inflammatory myopathies, and from the so-called fasciitis-panniculitis syndromes (Dalakas, 1991; Naschitz et al., 1996).
More than 130 patients with MMF have been recognized so far in France, and isolated cases have been recorded in the USA, the UK, Germany, Portugal and Spain (Cabello et al., 1999; Navarro et al., 1999). The aetiology of MMF has not been determined, but an environmental cause seems likely. On one hand, muscle lesions were repeatedly attributed to Whipple's disease (Misbah et al., 1997; Helliwell et al., 2000), and symptoms improved under antibiotic therapy in a few patients (Chérin et al., 1999), suggesting an infectious disease. However, polymerase chain reaction-based detection of Tropheryma whippeli and mycobacteria gave equivocal results in our laboratory (L. Belec, unpublished data). On the other hand, four of 18 patients from the initial series had ultrastructural evidence of small intracytoplasmic osmiophilic spiculated inclusions in macrophages (Gherardi et al., 1998). These inclusions resembled apatite crystals but were unstained by calcium stains (Gherardi et al., 1998). Their significance was unknown.
In the present study, we demonstrate that intracytoplasmic osmiophilic inclusions are constantly detected in MMF, and represent aluminium hydroxide crystals corresponding to the adjuvant used in some vaccines administered intramuscularly. Long-term persistence of aluminium hydroxide-associated inflammatory lesions at the injection site was detected in patients with diffuse myalgias that appeared subsequently to aluminium-containing vaccine administration.
Patients and methods
Inclusion criteria
All patients with MMF detected from 1993 to August 31, 1999, in the myopathological centres of Bordeaux, Créteil and Paris (Institut de Myologie) were included. MMF was identified at deltoid muscle biopsy by the presence of stereotyped sheets of densely packed non-epithelioid macrophages with a finely granular PAS-positive content, in epi-, peri- or endomysium.
Light and electron microscopy
Patients with MMF
Conventional light microscopy examination of muscle biopsy was performed in all patients, as previously described (Gherardi et al., 1998). In addition, 40 consecutive patients with MMF underwent ultrastructural examination of the infiltrate. Seven patients had infiltrates in the epoxy-embedded material, and 33 in the paraffin-embedded material. When necessary, paraffin was withdrawn in toluene and ethanol, before processing for epoxy-embedding and electron microscopy.
Controls
We compared the results with electron microscope examination of deltoid muscle biopsies from two groups of patients with well-characterized myopathic diseases collected in the same centre over the past 10 years: 40 adult patients with dermatomyositis at light microscopy (the most frequent idiopathic inflammatory myopathy in France), and 40 adult patients with muscle dystrophy of the facio-scapulo-humeral, Becker or limb girdle types.
X-ray microanalysis
Ultrathin sections of muscle specimens from eight patients were deposited on copper grids and coated with carbon for X-ray microanalysis. Analysis was carried out using an analytical transmission electron microscope and an energy dispersion spectrometer. Intracytoplasmic inclusions subjected to high energy electron beams emitted X-ray spectra showing series of narrow peaks specific to the emitting elements.
Nuclear microanalysis
Mapping and quantitative microanalysis were performed using a procedure analysing radiations emitted from the interaction of a proton beam with the matter (Moretto, 1996). Thick cryostat sections of muscle specimens from two patients were mounted on fresh formvar films, kept in the cryostat for 6 h and stored under silica gel. Mineral and metal ions were detected using the nuclear microprobe of the Centre d'Etudes Nucléaires de Bordeaux-Gradignan (Moretto, 1996). A 1 MeV proton beam focused down to a 2 μm spot was scanned over the sections in regions where infiltrates were observed on adjacent sections stained by haematoxylin-eosin. Particle induced X-ray emission and Rutherford backscattering spectrometry analyses were employed simultaneously and quantitative results were computed, as previously described (Moretto, 1996).
Atomic absorption spectrometry
The content of aluminium in dried muscle tissue was determined in an additional set of four patients with MMF (three samples including the lesion, four remote from the lesion) and in 14 normal controls. Aluminium plasma levels were determined in 20 MMF patients. Aluminium concentration was measured in duplicate by graphite furnace atomic absorption using a Zeeman-correction equipped 4100ZL spectrometer (Perkin-Elmer, Norwalk, Conn., USA). Stringent conditions were followed to avoid environmental contamination at all steps of sample processing (Pineau et al., 1993). Both internal and external quality control samples were used. The atomic absorption spectrometry laboratory participates in a worldwide interlaboratory aluminium quality control coordinated by the French Society of Clinical Biology (Poitiers, France).
Antibody testing
Antibody testing was performed in sera of 20 patients with MMF kept frozen at –80°C. IgG antibodies to tetanus toxoid (TT) were detected by immunoelectrophoresis (Hydragel-IEP; Sanofi Diagnostics-Pasteur, Marnes-la-Coquette, France). Screening for IgG to hepatitis A virus (HAV) and to hepatitis B virus (HBV) antigens (hepatitis B surface antigen HBsAg, hepatitis B core antigen HBcAg), and detection of HBsAg were performed using the IMX® System (Abbott Laboratories, Chicago, Ill., USA).
Immunization against HBsAg induces production of anti-HBs antibodies without anti-HBc antibodies and HBsAg, a profile typical of HBV vaccination.
Avidity of IgG antibodies to HAV was evaluated according to the dissociation of immune complexes by chaotropic ions in all HAV-positive MMF patients, 12 healthy subjects vaccinated against HAV 1 year prior to serum sampling and 23 patients previously infected by HAV. Briefly, 50 μl of serum were incubated with 150 μl of 1 M guanidium thiocyanate, or with 150 μl phosphate-buffered saline for 30 min at 37°C, and further processed for IgG to HAV detection. The relative avidity index was calculated as the ratio of the signal obtained with guanidium thiocyanate and that obtained with phosphate-buffered saline. Since the IMX® System for detection of IgG to HAV is a competitive assay, the relative avidity index is proportional to the avidity of antibodies for HAV antigens.
Clinicopathological correlations
We first compared the prevalence of myalgias in patients with and without MMF lesions who underwent deltoid muscle biopsy in the three participating centres. Only those data noted in files at time of biopsy were taken into account to consider patients as myalgic or non-myalgic.
Then, we re-evaluated MMF patients followed in our centres. Emphasis was placed on history of immunization with aluminium-containing vaccines, namely all HBV and HAV, and most TT vaccines. Immunization was assessed on the grounds of individual vaccination booklets and general practitioner files. Onset of myalgias, the most frequent symptom of MMF, was used to establish chronology of events. Patients were independently contacted by participating centres, and by the Institut de Veille Sanitaire (Saint-Maurice, France). In case of discrepancy between these sources, the patient was evaluated again by participating centres.
MMF rate of detection in vaccinated patients
This was assessed prospectively in Créteil and Bordeaux over a 1-year period (1999/2000). All patients undergoing a routine deltoid muscle biopsy in the non-dominant arm for investigation of a neuromuscular disorder in these two centres, were asked whether they had been immunized with aluminium hydroxide-containing vaccines within 8 years prior to biopsy. Muscle biopsy of immunized patients was categorized as MMF+ or MMF–. Delay from last immunization to biopsy was determined in each group on the basis of the vaccination booklet.
Intramuscular injection of vaccines in rats
Injection of an aluminium hydroxide-containing vaccine (GenHevac®, 250 μl) was performed into the tibialis anterior muscles of four adult Sprague–Dawley rats. Animals were sacrificed at 7, 14, 21 and 28 days post-injection, and the muscles close and remote from the site of injection were processed for optic and electron microscopy.
Statistical analysis
Results were expressed as mean ± standard error of the mean. Comparisons of biological results were performed using Student's t-test. Comparison of the prevalence of myalgias in patients with and without MMF lesions was performed using Fisher's exact test. Comparison of the delay from immunization to biopsy in MMF+ and MMF– patients was performed using the Mann–Whitney test.
Results
Patients
Altogether, 46 patients with MMF were detected in Bordeaux (20), Créteil (17) and Paris (nine), including six from 1993 to 1996, and 40 from 1997 to August 1999 (five in 1997, 12 in 1998 and 23 in 1999). From 1997 to August 1999, 34 out of 40 patients with MMF versus 565 out of 1252 patients without MMF had myalgias at time of deltoid muscle biopsy (P < 0.0001) (Table 1).
Eight additional patients were referred to the centres of Créteil (five) and Paris (three), making a total of 54 MMF patients followed by the participating centres. Information on immunizations was obtained for 50 of these MMF patients, including 48 adults and two children (12 and 14 years) (Table 2).
Light microscopy
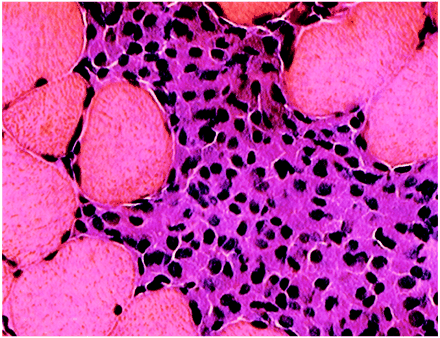
Stereotyped macrophage infiltrates were constantly (50 out of 50 patients) intermingled with lymphocytes (+ 28 out of 50, 56%; ++ 14 out of 50, 28%; +++ eight out of 50, 16%). Most lymphocytes were CD3+, and usually CD8+, T cells. Involvement of B cells was assessed by lymphoid follicle formation (11 out of 50, 22%) or presence of plasma cells (19 out of 50, 38%). Eosinophils (10%) and mast cells (10%) were occasionally observed. Muscle fibre damage was inconspicuous. HLA class I antigen was expressed by CD68+ macrophages and by muscle fibres located in the vicinity of the MMF lesion (Fig. 1).
Electron microscopy
Typical intracytoplasmic crystalline inclusions were detected in 40 out of 40 MMF cases and 0 out of 80 controls with dermatomyositis or muscle dystrophy. Inclusions appeared as aggregates of fine needle-shaped randomly oriented dense structures, forming clusters, often bounded by a distinct lysosomal membrane, in macrophages (Fig. 2). Extracellular deposits and microbial structures were not observed.
Microanalytical studies
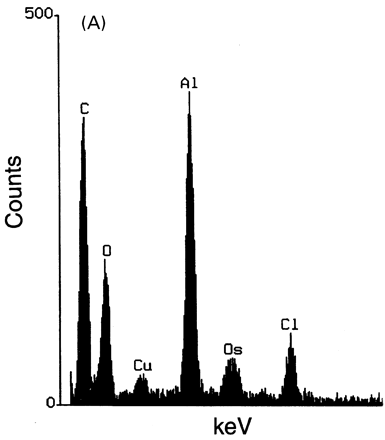
X-ray microanalysis of inclusions detected aluminium in eight out of eight cases (e.g. Fig. 3A). Nuclear microanalysis showed abundant aluminium in macrophages but not in myofibres (Fig. 3B). Phosphorus, present in both sites, was found at higher concentrations in macrophages. Other unexpected minerals or metals were not detected.
Atomic absorption spectrometry
Aluminium muscle levels were elevated (P < 0.0001), and higher in samples with, than without, macrophage infiltrates (P < 0.04). In contrast, all 20 sera from MMF patients had circulating aluminium levels below the normal limit (Table 3).
Antibody testing
All 20 sera from MMF patients had circulating antibodies against HBV (13 out of 20), HAV (14 out of 20) or TT (five out of 20). All positive HBV serologies (13 out of 13)were restricted to anti-HBs antibodies assessing previous immunization against HBV. Avidity index of anti-HAV antibodies was 5.00 ± 0.52, i.e. intermediate between the high index of previously infected individuals (7.45 ± 0.70) and the low index of recently vaccinated individuals (4.08 ± 0.59). Five HAV-seropositive MMF patients (five out of 14, 36%) had an avidity index of anti-HAV antibodies within the 95% confidence interval of avidity index of vaccinated patients.
Previous vaccinations in patients with MMF
Immunization against HBV had been performed prior to muscle biopsy in 42 out of 50 (84%) MMF patients. Routinely performed serology was consistent with such an immunization in 24 out of 26 of them. Anti-HBs antibody titres were within usual values (519 ± 111 IU/ml). The type of HBV vaccine administered was determined in 25 out of 42 patients: Engerix® (10); GenHevac B® (eight); both GenHevac B® (five) or Hevac B® (two) and Engerix® (seven) (Tables 2 and 4).
Immunization against HAV had been performed prior to muscle biopsy in seven out of 37 (19%) MMF patients in whom this could be determined. Routine HAV serology was positive in four out of four of them.
There was history of TT vaccine administration within 10 years previous to muscle biopsy in 46 out of 50 MMF patients: 12 had received TT vaccines that do not contain aluminium (DTP® or TP®), 22 had received TT vaccines that contain aluminium (Table 4) and 12 had received TT vaccines of an unknown type. Thus, 22 out of 38 (58%) patients in whom this could be determined had received aluminium-containing TT vaccines.
As a whole, there was definite evidence of previous immunization by one or more aluminium-containing vaccine in 50 out of 50 patients, including HBV vaccine alone (13), TT vaccine alone (eight) and various combinations of HBV vaccine with TT vaccine and/or HAV vaccine (29). MMF patients received one to nine (median four) doses of aluminium-containing vaccines within 10 years previous to biopsy. Delay from last vaccination to biopsy ranged from 3 months to 8 years (median 36 months).
There were diffuse myalgias in 47 out of 50 (94%) patients. Aluminium-containing vaccine administration was carried out prior to onset (44 patients) or worsening (two patients) of myalgias (46 out of 47, 98%). Thirty per cent of patients developed myalgias within 3 months after immunization, 61% within 1 year and 80% within 2 years. Thirty-four per cent of MMF patients also had a concurrent autoimmune disease (Table 2).
MMF rate of detection in vaccinated patients
Over 1 year, 113 patients with various neuromuscular disorders and previous immunization with aluminium-containing vaccines underwent a deltoid muscle biopsy in Créteil and Bordeaux: 97 (87%) had no detectable MMF lesions, and 16 (13%) had. Delay from immunization to biopsy could be established on the basis of the vaccination booklet in the 16 MMF+ patients and in 81 MMF– patients. The status MMF+ or – could not be attributed to a difference in the delay from immunization to biopsy, this delay being strictly similar in both groups (MMF+ range 12–96 months, median 42 months; MMF– range 3–96, median 42; MMF+ versus MMF–P = n.s.). All prospectively detected MMF+ patients but one had typical arthromyalgias and chronic fatigue (15 out of 16, 94%).
Intramuscular injection of vaccines in rats
Aluminium hydroxide-containing HBV vaccine induced a large necrotic area containing damaged muscle fibres and neutrophils, surrounded by abundant lymphocytes and macrophages (Day 7 and 15), that progressed to a mature lesion (Day 21 and 28), consisting of focal infiltration of densely packed large finely granular PAS-positive macrophages in endomysium, without giant cell formation or muscle fibre damage, very similar to the macrophage infiltrate of MMF (Fig. 4). Electron microscopy disclosed osmiophilic crystalline inclusions similar to those of MMF. Remote muscle was normal.
Discussion
In the present study, intracytoplasmic inclusions were constantly detected in macrophages of the MMF lesion, and were shown to contain aluminium by three different methods. Their crystalline structure was suggestive of aluminium oxyhydroxide (boehmite) rather than aluminium phosphate (Shirodkar et al., 1990). However, high levels of phosphorus were found in aluminium-loaded macrophages. Since phosphate anions generated by acid phosphatase activity in lysosomes are potent complexors of ionized aluminium, it is likely that a proportion of aluminium phosphate or hydroxyphosphate was formed in situ.
Aluminium intoxication causing encephalopathy, osteomalacia and anaemia has been mainly reported in patients with chronic renal failure undergoing haemodialysis (Salusky et al., 1991). Our patients had normal renal function tests (Gherardi et al., 1998), and their aluminium plasma levels were normal. This was inconsistent with passive aluminium deposition from blood, since aluminium tissue concentrations can usually be inferred from blood concentrations (Salusky et al., 1991; Flarend et al., 1997). Macrophage infiltrates were not observed anywhere else, other than in muscle (Gherardi et al., 1998), and the MMF lesion was exclusively detected in the deltoid muscle in adults. This reminded us that deltoid muscle is an elective site for vaccine injection, and that some vaccines may contain aluminium, an adjuvant frequently used to potentiate the immune response to vaccine antigens (Glenny et al., 1926).
In France, aluminium hydroxide is found in HBV, HAV and most TT vaccines. Elsewhere, it may be found in additional vaccines, such as the six-shot anthrax vaccine administered to the US military personnel (Product Insert for Anthrax Vaccine, 2001). Retrospective serological analysis was either consistent or compatible with previous HBV, HAV or TT immunization in all tested MMF patients. Prevalence of HBV immunization was much higher in these patients (65%) than in the French population of similar age (16–22% in 1996).
History revealed that all MMF patients (50 out of 50) had been immunized 3 months to 8 years before muscle biopsy by aluminium-containing vaccines, including HBV (26%), TT (16%) or combinations of HBV, TT and HAV (58%) vaccines. Rats injected intramuscularly with an aluminium hydroxide-containing vaccine developed infiltrates of macrophages with intracytoplasmic crystalline inclusions very similar to those of MMF. Taken together with previous reports on granulomas induced by various aluminium-containing compounds in humans and animals (Balouet et al., 1977; Gotto and Akama, 1982; Mrak, 1982; Miliauskas et al., 1993; Garcia-Patos et al., 1995), these results firmly establish that aluminium hydroxide-containing vaccines represent the direct cause of the MMF lesion.
The onset of MMF detection was probably related to introduction of the intramuscular route of vaccination in the early 1990s. The striking number of cases detected in France, compared with other countries, is more puzzling. A local pharmaceutical cause linked to manufacturing practices seems unlikely (WHO Vaccine Safety Advisory Committee, 1999). Three combined factors might explain why MMF has been mainly detected in France: (i) MMF probably came to medical attention in France because of extensive immunization programs carried out in this country; (ii) a large number of French people have been primovaccinated at adulthood against hepatitis B which is not the case in other countries, where HBV immunization is restricted to children and risk groups; and (iii) deltoid muscle is a biopsy site more commonly used in France than in other countries (WHO Vaccine Safety Advisory Committee, 1999).
Once injected into tissues, aluminium hydroxide forms a deposit, damages the injected tissue, elicits danger signals from stressed cells, attracts inflammatory and antigen-presenting cells and is subjected to phagocytosis (World Health Organization, 1976; Balouet et al., 1977; Schijns, 2000). Phagocytosed aluminium hydroxide increases survival of macrophages and synergizes the effects of M-CSF (monocyte-colony stimulating factor) and GM-CSF (granulocyte/monocyte colony stimulating factor) (Hamilton et al., 2000). A number of aluminium-loaded macrophages accumulate locally while others migrate to the regional lymph node (World Health Organization, 1976). Aluminium hydroxide induces monocyte-derived cells to acquire the phenotype and APC (antigen presenting cell) functions of dendritic cells (Ulanova et al., 2001). Antigen presentation at the injection site is assessed by the presence of lymphoplasmacytic infiltrates in the vicinity of macrophages, defining the so-called immunogenic granuloma that is not formed when the adjuvant is used alone, and that increases when polyantigens are added to the aluminium-adjuvanted vaccine (Balouet et al., 1977). It is currently believed that the injected aluminium adjuvant is progressively solubilized into blood, redistributed to tissues and gradually secreted into urine (Flarend et al., 1997). Using 26Al-labelled adjuvants, Flarend and colleagues showed that 17% of a full human dose of aluminium hydroxide, and 51% of a similar dose of aluminium phosphate are eliminated in urine within 28 days after intramuscular injection into rabbits (Flarend et al., 1997). Residence time of aluminium hydroxide in muscle has not been established in spite of its long use in vaccines. A recent study in rabbits showed that macrophage accumulation develops in all injected sites, decreases by day 30, and disappears from most sites within 3 months post-injection (Francıois Verdier, Aventis-Pasteur, France, presentation at the `Aluminium in vaccines' CDC symposium held in San Juan, Puerto Rico, May 11 and 12, 2000; personal communication). In our laboratory, a residence time longer than 6 months has been observed in rats (Authier et al., 2001a). Further investigations on this important topic are desirable.
Ethical reasons prevented us from performing muscle biopsies in healthy individuals to estimate the residence time of aluminium hydroxide in the human muscle. However, a prospective evaluation showed that MMF detection at deltoid muscle biopsy is rare among aluminium hydroxide-containing vaccine recipients with neuromuscular disorders. Time elapsed from vaccination to histological detection of MMF could be very long, i.e. up to 8 years, the mean delay being 36 months. Both findings, and the previously reported intersubject variability in elimination of aluminium (Balouet et al., 1977; Talbot et al., 1995; Flarend et al., 1997), led the WHO Vaccine Safety Advisory Committee to propose as a working hypothesis that MMF could occur in `a predisposed subset of individuals with impaired ability to clear aluminium from the deltoid muscle' (WHO Vaccine Safety Advisory Committee, 1999).
Clinical manifestations of patients with MMF mainly include steroid responsive diffuse myalgias, arthralgias and chronic fatigue (Gherardi et al., 1998; Chérin et al., 2000). Typically, patients have myalgias of a localized onset, most often in lower limbs, with subsequent extension leading to diffuse muscle pain (Institut de Veille Sanitaire, 2001). Prevalence of myalgias was much higher in MMF patients than in the other 1252 patients who had a deltoid muscle biopsy at the same time in the same centres. Myalgia onset was subsequent to vaccination in 94% of MMF patients and, as a rule, other causes of myalgia were not detected. Taken together, these data make fortuitous association of MMF with chronic myalgias very unlikely. Myalgias, arthralgias and fatigue are among the most common post-vaccinal symptoms reported to passive surveillance systems (Vaccine Adverse Reaction Searchable Database, 2000). Myalgias and arthralgias already have been recognized as adverse effects of HBV vaccination (McMahon et al., 1992), and a possible association between chronic fatigue and HBV vaccination has been debated (Working Group on the Possible Relationship between Hepatitis B Vaccination and the Chronic Fatigue Syndrome, 1993). Most MMF patients had their first myalgias several months after vaccination, and would have not been considered to have an adverse reaction to vaccine using standard criteria (Tourbah et al., 1999). In light of the possible occurrence of long-term persistence of vaccine compounds in the injected tissues, we feel that the short post-exposure period during which imputability of symptoms to an adverse drug reaction is usually accepted might be inappropriate in the setting of vaccines.
Most chronic myalgia and fatigue syndromes remain poorly understood. Patients with MMF had a myopathic electromyogram (42%), CK (creatine kinase) elevation (50%) and abnormal 67Ga scintigraphy (100%) showing increased gallium global uptake predominating in painful areas along limb muscle fascias and in para-articular tissues (Chérin et al., 2000). Significance of such a gallium uptake in MMF patients is at present unclear. A few patients had a second muscle biopsy remote from the MMF detection site, showing poorly specific inflammatory infiltrates and no evidence of MMF diffusion (data not shown). It is not unprecedented that a focal muscle stimulus may elicit diffuse myalgias of an unknown cause: in a recent experimental study, bilateral, long-lasting hyperalgesia was induced by unilateral intramuscular injections of acidic saline, in the absence of histological muscle lesions (Sluka et al., 2001).
Both HBV and TT vaccines were implicated in our patients, suggesting a role of the adjuvant in the vaccine-associated systemic effects (McMahon et al., 1992). In addition to deposit formation, aluminium hydroxide potently stimulates the immune system (Brewer et al., 1999). Aluminium hydroxide has been reported to induce IL-1 (interleukin-1) production by monocytes, complement activation, eosinophilia, increased specific and non-specific IgG1 and IgE antibody responses and delayed-type hypersensitivity (Gupta et al., 1995). In the present series, MMF lesions constantly included a lymphoid component, ranging from lymphoplasmacytic infiltrates to organized tertiary lymphoid tissue, assessing an ongoing immunological process at time of biopsy. Persistent systemic immune activation that fails to `switch off' previously has been regarded as the possible cause of chronic fatigue and arthromyalgias (Landay et al., 1991; Hassan et al., 1998), through sustained release of inflammatory cytokines and production of autotoxic T cells and autoantibodies (Konstantinov et al., 1996). Consistently, we have observed that MMF patients have B-cell hyperlymphocytosis, higher IL-6 circulatory levels than healthy vaccinated controls and detectable circulating antinuclear and anti-phospholipid autoantibodies (50%) (Gherardi et al., 2001). These data indicate that MMF is associated with a shift of immune responses towards a Th-2 profile, which is typically induced by aluminium hydroxide (Brewer et al., 1999), and probably contributes to emergence of chronic fatigue and associated manifestations (Rook and Zumla, 1997).
Patients with MMF may also have co-existent autoimmune diseases (Authier et al., 2001b). The significance of this remains uncertain, but aluminium potently induces oxidative stress and lipid peroxidation (Gutteridge et al., 1985; Xie et al., 1996; Yoshino et al., 1999; Campbell and Bondy, 2000), which may reveal cryptic immunogenic epitopes (Horkko et al., 1996; Casciola-Rosen et al., 1997; Petrovas et al., 1999; Kalluri et al., 2000). Metals in a suitable microenvironment could also favour activation of autoreactive T cells that exist in healthy individuals (Fournie et al., 2001). Whether such mechanisms of autoimmunity are involved in MMF patients deserves further investigation.
At this point, an epidemiological survey aimed at evaluating the putative link between long-term persistence of MMF lesions at sites of vaccine injection and systemic symptoms is required. Meanwhile, there is no basis for recommending a change in HBV vaccination practices (WHO Vaccine Safety Advisory Committee, 1999).
We conclude that: (i) intracytoplasmic inclusions are constantly detected in MMF lesions and correspond to aluminium hydroxide crystals; (ii) MMF lesions are secondary to intramuscular injections of aluminium hydroxide-containing vaccines and should be regarded as a post-vaccinal immunogenic granuloma; and (iii) MMF lesions are usually detected in the deltoid muscle of patients with diffuse myalgias appearing subsequently to aluminium hydroxide-containing vaccine administration.
Prevalence of myalgias in patients with a deltoid muscle biopsy performed from 1997 to August 1999
| . | Macrophagic myofasciitis [n (%)] . | Other [n (%)] . |
|---|---|---|
| P < 0.0001 by Fisher's exact test. | ||
| Myalgias | 34 (85) | 565 (45) |
| No myalgias | 6 (15) | 687 (55) |
| Total | 40 (100) | 1252 (100) |
| . | Macrophagic myofasciitis [n (%)] . | Other [n (%)] . |
|---|---|---|
| P < 0.0001 by Fisher's exact test. | ||
| Myalgias | 34 (85) | 565 (45) |
| No myalgias | 6 (15) | 687 (55) |
| Total | 40 (100) | 1252 (100) |
Prevalence of myalgias in patients with a deltoid muscle biopsy performed from 1997 to August 1999
| . | Macrophagic myofasciitis [n (%)] . | Other [n (%)] . |
|---|---|---|
| P < 0.0001 by Fisher's exact test. | ||
| Myalgias | 34 (85) | 565 (45) |
| No myalgias | 6 (15) | 687 (55) |
| Total | 40 (100) | 1252 (100) |
| . | Macrophagic myofasciitis [n (%)] . | Other [n (%)] . |
|---|---|---|
| P < 0.0001 by Fisher's exact test. | ||
| Myalgias | 34 (85) | 565 (45) |
| No myalgias | 6 (15) | 687 (55) |
| Total | 40 (100) | 1252 (100) |
Characteristics of the 50 patients with macrophagic myofasciitis
| Characteristic . | Value . |
|---|---|
| Age (years) | |
| Median | 46 |
| Range | 12–77 |
| Sex [n (%)] | |
| Male | 26/50 (52) |
| Female | 24/50 (48) |
| Aluminium hydroxide-containing vaccine administration [n (%)] | |
| Overall number | 50/50 (100) |
| HBV-vaccine | 42/50 (84) |
| HAV-vaccine | 7/37 (19) |
| TT-vaccine (within 10 years) | 22/38 (58) |
| Number of doses per patient | |
| Median | 4 |
| Range | 1–9 |
| Delay from vaccination to biopsy (months) | |
| Median | 36 |
| Range | 3–96 |
| Overall number of myalgias [n (%)] | 47/50 (94) |
| Delay from vaccination to myalgias | |
| Myalgia onset after vaccination [n (%)] | 44/47 (94) |
| Myalgia increase after vaccination [n (%)] | 2/47 (4) |
| Median (months) | 11 |
| Range (months) | 0–72 |
| ≤3 months [n (%)] | 14/46 (30) |
| ≤1 year [n (%)] | 28/46 (61) |
| ≤2 years [n (%)] | 37/46 (80) |
| Concurrent autoimmune disease [n (%)] | |
| Overall number | 17/50 (34) |
| Multiple sclerosis | 6/50 (12) |
| Inclusion body myositis | 3/50 (6) |
| Dermatomyositis | 2/50 (4) |
| Hashimoto's thyroiditis | 2/50 (4) |
| Rheumatoid arthritis | 2/50 (4) |
| Other | 2/50 (4) |
| Characteristic . | Value . |
|---|---|
| Age (years) | |
| Median | 46 |
| Range | 12–77 |
| Sex [n (%)] | |
| Male | 26/50 (52) |
| Female | 24/50 (48) |
| Aluminium hydroxide-containing vaccine administration [n (%)] | |
| Overall number | 50/50 (100) |
| HBV-vaccine | 42/50 (84) |
| HAV-vaccine | 7/37 (19) |
| TT-vaccine (within 10 years) | 22/38 (58) |
| Number of doses per patient | |
| Median | 4 |
| Range | 1–9 |
| Delay from vaccination to biopsy (months) | |
| Median | 36 |
| Range | 3–96 |
| Overall number of myalgias [n (%)] | 47/50 (94) |
| Delay from vaccination to myalgias | |
| Myalgia onset after vaccination [n (%)] | 44/47 (94) |
| Myalgia increase after vaccination [n (%)] | 2/47 (4) |
| Median (months) | 11 |
| Range (months) | 0–72 |
| ≤3 months [n (%)] | 14/46 (30) |
| ≤1 year [n (%)] | 28/46 (61) |
| ≤2 years [n (%)] | 37/46 (80) |
| Concurrent autoimmune disease [n (%)] | |
| Overall number | 17/50 (34) |
| Multiple sclerosis | 6/50 (12) |
| Inclusion body myositis | 3/50 (6) |
| Dermatomyositis | 2/50 (4) |
| Hashimoto's thyroiditis | 2/50 (4) |
| Rheumatoid arthritis | 2/50 (4) |
| Other | 2/50 (4) |
Characteristics of the 50 patients with macrophagic myofasciitis
| Characteristic . | Value . |
|---|---|
| Age (years) | |
| Median | 46 |
| Range | 12–77 |
| Sex [n (%)] | |
| Male | 26/50 (52) |
| Female | 24/50 (48) |
| Aluminium hydroxide-containing vaccine administration [n (%)] | |
| Overall number | 50/50 (100) |
| HBV-vaccine | 42/50 (84) |
| HAV-vaccine | 7/37 (19) |
| TT-vaccine (within 10 years) | 22/38 (58) |
| Number of doses per patient | |
| Median | 4 |
| Range | 1–9 |
| Delay from vaccination to biopsy (months) | |
| Median | 36 |
| Range | 3–96 |
| Overall number of myalgias [n (%)] | 47/50 (94) |
| Delay from vaccination to myalgias | |
| Myalgia onset after vaccination [n (%)] | 44/47 (94) |
| Myalgia increase after vaccination [n (%)] | 2/47 (4) |
| Median (months) | 11 |
| Range (months) | 0–72 |
| ≤3 months [n (%)] | 14/46 (30) |
| ≤1 year [n (%)] | 28/46 (61) |
| ≤2 years [n (%)] | 37/46 (80) |
| Concurrent autoimmune disease [n (%)] | |
| Overall number | 17/50 (34) |
| Multiple sclerosis | 6/50 (12) |
| Inclusion body myositis | 3/50 (6) |
| Dermatomyositis | 2/50 (4) |
| Hashimoto's thyroiditis | 2/50 (4) |
| Rheumatoid arthritis | 2/50 (4) |
| Other | 2/50 (4) |
| Characteristic . | Value . |
|---|---|
| Age (years) | |
| Median | 46 |
| Range | 12–77 |
| Sex [n (%)] | |
| Male | 26/50 (52) |
| Female | 24/50 (48) |
| Aluminium hydroxide-containing vaccine administration [n (%)] | |
| Overall number | 50/50 (100) |
| HBV-vaccine | 42/50 (84) |
| HAV-vaccine | 7/37 (19) |
| TT-vaccine (within 10 years) | 22/38 (58) |
| Number of doses per patient | |
| Median | 4 |
| Range | 1–9 |
| Delay from vaccination to biopsy (months) | |
| Median | 36 |
| Range | 3–96 |
| Overall number of myalgias [n (%)] | 47/50 (94) |
| Delay from vaccination to myalgias | |
| Myalgia onset after vaccination [n (%)] | 44/47 (94) |
| Myalgia increase after vaccination [n (%)] | 2/47 (4) |
| Median (months) | 11 |
| Range (months) | 0–72 |
| ≤3 months [n (%)] | 14/46 (30) |
| ≤1 year [n (%)] | 28/46 (61) |
| ≤2 years [n (%)] | 37/46 (80) |
| Concurrent autoimmune disease [n (%)] | |
| Overall number | 17/50 (34) |
| Multiple sclerosis | 6/50 (12) |
| Inclusion body myositis | 3/50 (6) |
| Dermatomyositis | 2/50 (4) |
| Hashimoto's thyroiditis | 2/50 (4) |
| Rheumatoid arthritis | 2/50 (4) |
| Other | 2/50 (4) |
Aluminium concentrations in muscle tissue and plasma
| Sample . | Aluminium range (mean ± SEM) . |
|---|---|
| Muscle tissue | |
| MMF: macrophage infiltrate (n = 3) | 77–1428 mg/g (584 ± 425) |
| MMF: remote from macrophage infiltrate (n = 4) | 5–431 mg/g (137 ± 100) |
| Normal muscle (n = 14) | 1–58 mg/g (10 ± 5) |
| Plasma | |
| MMF (n = 20) | 0.1–0.4 mM |
| Normal values | 0.1–0.4 mM |
| Sample . | Aluminium range (mean ± SEM) . |
|---|---|
| Muscle tissue | |
| MMF: macrophage infiltrate (n = 3) | 77–1428 mg/g (584 ± 425) |
| MMF: remote from macrophage infiltrate (n = 4) | 5–431 mg/g (137 ± 100) |
| Normal muscle (n = 14) | 1–58 mg/g (10 ± 5) |
| Plasma | |
| MMF (n = 20) | 0.1–0.4 mM |
| Normal values | 0.1–0.4 mM |
Aluminium concentrations in muscle tissue and plasma
| Sample . | Aluminium range (mean ± SEM) . |
|---|---|
| Muscle tissue | |
| MMF: macrophage infiltrate (n = 3) | 77–1428 mg/g (584 ± 425) |
| MMF: remote from macrophage infiltrate (n = 4) | 5–431 mg/g (137 ± 100) |
| Normal muscle (n = 14) | 1–58 mg/g (10 ± 5) |
| Plasma | |
| MMF (n = 20) | 0.1–0.4 mM |
| Normal values | 0.1–0.4 mM |
| Sample . | Aluminium range (mean ± SEM) . |
|---|---|
| Muscle tissue | |
| MMF: macrophage infiltrate (n = 3) | 77–1428 mg/g (584 ± 425) |
| MMF: remote from macrophage infiltrate (n = 4) | 5–431 mg/g (137 ± 100) |
| Normal muscle (n = 14) | 1–58 mg/g (10 ± 5) |
| Plasma | |
| MMF (n = 20) | 0.1–0.4 mM |
| Normal values | 0.1–0.4 mM |
Exposure of patients with macrophagic myofasciitis to aluminium-containing vaccines available in France
| Commercial name . | No. of patients . |
|---|---|
| *Withdrawn. | |
| HBV vaccines | |
| Engerix B20 adulte® | 17 |
| Vaccin GenHevac B Pasteur® | 13 |
| Vaccin Hevac B Pasteur®* | 2 |
| HBvax® | 0 |
| TT vaccines | |
| Tetavax® | 13 |
| Vaccin tétanique Pasteur® | 3 |
| D.T. Vax® | 2 |
| D.T. Coq® | 1 |
| Vaccin D.T.C.P. Pasteur® | 1 |
| Tetracoq® | 1 |
| Vaccin D.T. Pasteur®* | 0 |
| Vaxicoq® | 0 |
| HAV vaccines | |
| Vaccin Havrix® | 7 |
| Vaccin Twinrix adulte 720/20® | 0 |
| Vaccin Twinrix enfant 360/10® | 0 |
| Commercial name . | No. of patients . |
|---|---|
| *Withdrawn. | |
| HBV vaccines | |
| Engerix B20 adulte® | 17 |
| Vaccin GenHevac B Pasteur® | 13 |
| Vaccin Hevac B Pasteur®* | 2 |
| HBvax® | 0 |
| TT vaccines | |
| Tetavax® | 13 |
| Vaccin tétanique Pasteur® | 3 |
| D.T. Vax® | 2 |
| D.T. Coq® | 1 |
| Vaccin D.T.C.P. Pasteur® | 1 |
| Tetracoq® | 1 |
| Vaccin D.T. Pasteur®* | 0 |
| Vaxicoq® | 0 |
| HAV vaccines | |
| Vaccin Havrix® | 7 |
| Vaccin Twinrix adulte 720/20® | 0 |
| Vaccin Twinrix enfant 360/10® | 0 |
Exposure of patients with macrophagic myofasciitis to aluminium-containing vaccines available in France
| Commercial name . | No. of patients . |
|---|---|
| *Withdrawn. | |
| HBV vaccines | |
| Engerix B20 adulte® | 17 |
| Vaccin GenHevac B Pasteur® | 13 |
| Vaccin Hevac B Pasteur®* | 2 |
| HBvax® | 0 |
| TT vaccines | |
| Tetavax® | 13 |
| Vaccin tétanique Pasteur® | 3 |
| D.T. Vax® | 2 |
| D.T. Coq® | 1 |
| Vaccin D.T.C.P. Pasteur® | 1 |
| Tetracoq® | 1 |
| Vaccin D.T. Pasteur®* | 0 |
| Vaxicoq® | 0 |
| HAV vaccines | |
| Vaccin Havrix® | 7 |
| Vaccin Twinrix adulte 720/20® | 0 |
| Vaccin Twinrix enfant 360/10® | 0 |
| Commercial name . | No. of patients . |
|---|---|
| *Withdrawn. | |
| HBV vaccines | |
| Engerix B20 adulte® | 17 |
| Vaccin GenHevac B Pasteur® | 13 |
| Vaccin Hevac B Pasteur®* | 2 |
| HBvax® | 0 |
| TT vaccines | |
| Tetavax® | 13 |
| Vaccin tétanique Pasteur® | 3 |
| D.T. Vax® | 2 |
| D.T. Coq® | 1 |
| Vaccin D.T.C.P. Pasteur® | 1 |
| Tetracoq® | 1 |
| Vaccin D.T. Pasteur®* | 0 |
| Vaxicoq® | 0 |
| HAV vaccines | |
| Vaccin Havrix® | 7 |
| Vaccin Twinrix adulte 720/20® | 0 |
| Vaccin Twinrix enfant 360/10® | 0 |
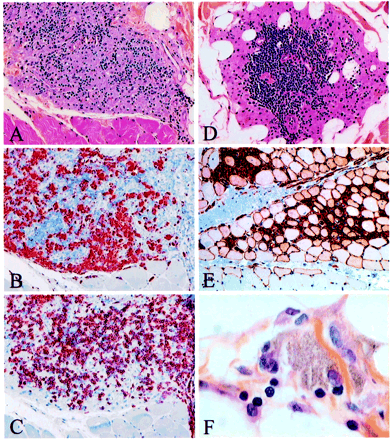
Deltoid muscle biopsy of patients with macrophagic myofasciitis (light microscopy): (A) tightly packed macrophages intermingled with lymphocytes in epimysium (haematoxylin and eosin, ×100); (B) adjacent section of the same biopsy showing immunolocalization of the macrophage marker CD68 (alkaline phosphatase–anti-alkaline phosphatase, APAAP, ×100); (C) adjacent section of the same biopsy showing immunolocalization of the T-cell marker CD3 (APAAP, ×100); (D) nodular aggregation of lymphocytes with microvascular neoangiogenesis, consistent with a primary lymphoid follicle developed in the centre of an epimysial islet of macrophages (haematoxylin and eosin, ×100); (E) HLA class I antigen expression in both macrophages accumulated in endomysium and adjacent muscle fibres (peroxidase–anti-peroxidase, PAP, ×70); and (F) lymphocytes stuck to a granular macrophage (haematoxylin and eosin, ×600).
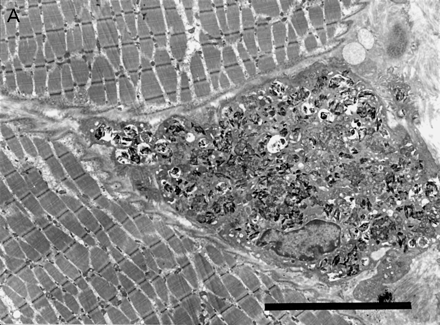
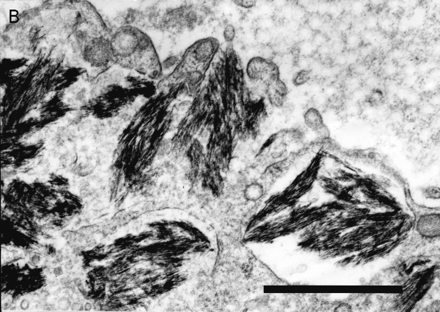
Deltoid muscle biopsy of patients with macrophagic myofasciitis (electron microscopy): (A) an endomysial macrophage filled with dense osmiophilic intracytoplasmic inclusions (bar = 10 μm); (B) at higher magnification inclusions are frequently membrane-bound and show a finely spicular structure (bar = 0.5 μm).
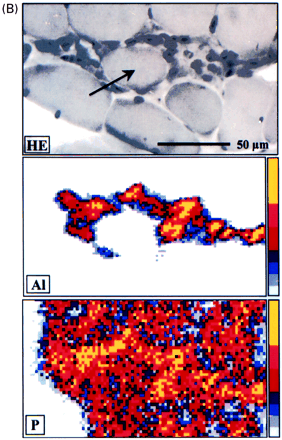
Microanalytic studies. (A) X-ray microanalysis: low energetic X-ray spectra obtained using energy dispersion spectrometer of intracytoplasmic inclusions of macrophages showing a peak specific of aluminium (Al) and peaks due to copper (Cu), osmium (Os) and chloride (Cl) that constitute the background. (B) Nuclear microanalysis: particle induced X-ray emission microanalysis showing an abnormal presence of aluminium in muscle tissue strictly restricted to the areas of macrophage infiltrates: aluminium level was 53 310 ± 9600 μg/g in macrophages and 105 ± 20 μg/g in the centre of the muscle fibre. A spatial correlation is observed on the elemental maps for aluminium and phosphorus.
Intramuscular injection site of HBV vaccine in a Sprague–Dawley rat at Day 21 post-injection: tightly packed basophilic macrophages infiltrated between muscle fibres (haematoxylin and eosin, ×250).
We are most indebted to Dr Philippe Malfait and to Dr Daniel Levy-Bruhl from the Institut de Veille Sanitaire (Saint-Maurice, France) for their help in the design of the study, and for their kind cooperation for extraction of vaccination data. Data and conclusions of the present paper have been analysed by the Safety of Vaccines Standing Committee of the World Health Organization (Geneva, Switzerland), by the Institut de Veille Sanitaire (Saint-Maurice, France) and Agence Francıaise de Sécurité Sanitaire des Produits de Santé (Saint-Denis, France) and by experts from the Centres of Disease Control (Atlanta, USA). This work was supported by the Association Francıaise contre les Myopathies (AFM).
References
Authier FJ, Sauvat S, Poron F, Gherardi RK. Experimental macrophagic myofasciitis: a long-term evaluation in different rat strains [abstract].
Authier FJ, Chérin P, Créange A, Bonnotte P, Ferrer X, Abdlelmoumni A, et al. Central nervous system disease in patients with macrophagic myofasciitis.
Balouet G, Baret M, Relyveld E, Ravisse P, Levaditi J. Rôle des antigènes et des substances adjuvantes dans la réponse histologique au cours des granulomes experimentaux (`granulome immunogène').
Brewer JM, Conacher M, Hunter CA, Mohrs M, Brombacher F, Alexander J. Aluminium hydroxide adjuvant initiates strong antigen-specific Th2 responses in the absence of IL-4 and IL-13-mediated signaling.
Cabello A, Ruiz-Falco ML, Garcia-Peña JJ, Madero S, Ferreiro A, Báez B, et al. Macrophagic myofasciitis: a new case of non-French origin [abstract].
Campbell A, Bondy SC. Aluminum-induced oxidative events and its relation to inflammation: a role for the metal in Alzheimer's disease.
Casciola-Rosen L, Wigley F, Rosen A. Scleroderma autoantigens are uniquely fragmented by metal-catalyzed oxidation reactions: implications for pathogenesis.
Chérin P, Gherardi RK, Pennaforte JL. Macrophagic myofasciitis: improvement with antibiotic therapy [letter]. J.
Chérin P, Authier FJ, Gherardi RK, Romero N, Lafôret P, Eymard B, et al. Gallium-67 scintigraphy in macrophagic myofasciitis.
Dalakas MC. Polymyositis, dermatomyositis and inclusion-body myositis. [Review].
Flarend RE, Hem SL, White JL, Elmore D, Suckow MA, Rudy AC, et al. In vivo absorption of aluminium-containing vaccine adjuvants using 26Al.
Fournie GJ, Mas M, Cavtain B, Savignac M, Subra JF, Pelletier L, et al. Induction of autoimmunity through bystander effects—lessons from immunological disorders induced by heavy metals.
Garcia-Patos V, Pujol RM, Alomar A, Cistero A, Curell R, Fernandez-Figueras MT, et al. Persistent subcutaneous nodules in patients hyposensitized with aluminum-containing allergen extracts.
Gherardi RK, Coquet M, Chérin P, Authier FJ, Laforêt P, Belec L, et al. Macrophagic myofasciitis: an emerging entity.
Gherardi RK, Plonquet A, André C, Intrator L, Poron F, Coquet M, et al. Macrophagic myofasciitis: evidence for chronic local and systemic immune activation associated with persistence of aluminum hydroxide-loaded macrophages in muscle [abstract].
Glenny AT, Pope CG, Waddington H, Wallace UJ. The antigenic value of toxoid precipitated by potassium alum.
Goto N, Akama K. Histopathological studies of reactions in mice injected with aluminum-adsorbed tetanus toxoid.
Gupta RK, Rost BE, Relyveld E, Siber GR. Adjuvant properties of aluminum and calcium compounds. In: Powell MF, Newman MJ, editors.Vaccine design: the subunit and adjuvant approach. New York: Plenum Press; 1995. p. 229–48.
Gutteridge JM, Quinlan GJ, Clark I, Halliwell B. Aluminium salts accelerate peroxidation of membrane lipids stimulated by iron salts.
Hamilton JA, Byrne R, Whitty G. Particulate adjuvants can induce macrophage survival, DNA synthesis, and a synergistic proliferative response to GM-CSF and CSF-1.
Hassan IS, Bannister BA, Akbar A, Weir W, Bofill M. A study of the immunology of the chronic fatigue syndrome: correlation of immunologic parameters to health dysfunction.
Helliwell TR, Appleton RE, Mapstone NC, Davidson J, Walsh KP. Dermatomyositis and Whipple's disease.
Horkko S, Miller E, Dudl E, Reaven P, Curtiss LK, Zvaifler NJ, et al. Antiphospholipid antibodies are directed against epitopes of oxidized phospholipids. Recognition of cardiolipin by monoclonal antibodies to epitopes of oxidized low density lipoprotein.
Institut de Veille Sanitaire. Myofasciite à macrophages: rapport d'investigation exploratoire. Mars 2001 [cited 2001 May 21]. Available from: URL: http://www.invs.sante.fr
Kalluri R, Cantley LG, Kerjaschki D, Neilson EG. Reactive oxygen species expose cryptic epitopes associated with autoimmune Goodpasture syndrome.
Konstantinov K, von Mikecz A, Buchwald D, Jones J, Gerace L, Tan EM. Autoantibodies to nuclear envelope antigens in chronic fatigue syndrome.
Landay AL, Jessop C, Lennette ET, Levy JA. Chronic fatigue syndrome: clinical condition associated with immune activation.
McMahon BJ, Helminiak C, Wainwright RB, Bulkow L, Trimble BA, Wainwright K. Frequency of adverse reactions to hepatitis B vaccine in 43,618 persons.
Miliauskas JR, Mukherjee T, Dixon B. Postimmunization (vaccination) injection-site reactions. A report of four cases and review of the literature. [Review].
Misbah SA, Ozols B, Franks A, Mapstone N. Whipple's disease without malabsorption: new atypical features.
Naschitz JE, Boss JH, Misselevich I, Yeshurun D, Rosner I. The fasciitis-panniculitis syndromes. Clinical and pathologic features. [Review].
Navarro C, Rodrìguez D, Willisch A, Fachal C, Teijero A, Fernandez R, et al. Macrophagic myofasciitis. A report of a new Spanish case [abstract].
Petrovas C, Vlachoyiannopoulos PG, Kordossis T, Moutsopoulos HM. Anti-phospholipid antibodies in HIV infection and SLE with or without anti-phospholipid syndrome: comparisons of phospholipid specificity, avidity and reactivity with beta2-GPI.
Pineau A, Guillard O, Chappuis P, Arnaud J, Zawislak R. Sampling conditions for biological fluids for trace elements monitoring in hospital patients: a critical approach. [Review].
Product Insert for Anthrax Vaccine [cited 2001 May 21]. Available from: URL: http://www.dallasnw.quik.com/cyberella/Anthrax/Product.html
Rook GA, Zumla A. Gulf War syndrome: is it due to a systemic shift in cytokine balance towards a Th2 profile?
Sluka KA, Kalra A, Moore SA. Unilateral intramuscular injections of acidic saline produce a bilateral, long-lasting hyperalgesia.
Salusky IB, Foley J, Nelson P, Goodman WG. Aluminum accumulation during treatment with aluminum hydroxide and dialysis in children and young adults with chronic renal disease.
Schijns VE. Immunological concepts of vaccine adjuvant activity. [Review].
Shirodkar S, Hutchinson RL, Perry DL, White JL, Hem SL. Aluminum compounds used as adjuvants in vaccines.
Talbot RJ, Newton D, Priest ND, Austin JG, Day JP. Inter-subject variability in the metabolism of aluminium following intravenous injection as citrate.
Tourbah A, Gout O, Liblau R, Lyon-Caen O, Bougniot C, Iba-Zizen MT, et al. Encephalitis after hepatitis B vaccination. Recurrent disseminated encephalitis or MS?
Ulanova M, Tarkowski A, Hahn-Zoric M, Hanson LA. The common vaccine adjuvant aluminum hydroxide up-regulates accessory properties of human monocytes via an interleukin-4-dependent mechanism.
Vaccine Adverse Event Reporting System (VAERS): searchable database [cited 2001 May 21]. Available from: URL: http://www.fedbuzz.com/vaccine/vac.html
WHO Vaccine Safety Advisory Committee. Macrophagic myofasciitis and aluminum-containing vaccines.
Working Group on the Possible Relationship between Hepatitis B Vaccination and the Chronic Fatigue Syndrome. Report.
World Health Organization. Immunological adjuvants. Technical Report Series 595. Geneva: World Health Organization; 1976. 40pp.
Xie CX, Mattson MP, Lovell MA, Yokel RA. Intraneuronal aluminum potentiates iron-induced oxidative stress in cultured rat hippocampal neurons.